Problems
- Details
- Last Updated: Wednesday, 26 March 2014 13:14
My simulation keeps crashing, what can I do?
The following is a list of suggestions that might help you stabilize your system:
Reduce the time step somewhat, some applications require 20 fs rather than 30-40 fs. If you need to go below 20 fs something else is likely to be wrong.
Increase the neighbourlist update frequency and/or neighborlist cutoff size.
Replace contraints by (stiff) bonds, recommended during minimization
Replace stiff bonds by constraints, this will increase stability and allow larger time steps. Stiff bonds are bonds with a force constant exceeding ~ 10000 kJ mol-1 nm-2.
For beta-strands, you might try using distance constraints, available as option in the itp-generating script. If proteins keep crashing in general, try adding an elastic network to your protein, using ELNEDYN.
Play with your topology, you might have conflicting bonded potentials. Also make sure you have the appropriate exclusions (nearest neighbours should alway be excluded, but sometimes 2nd or 3rd nearest neighbours as well.
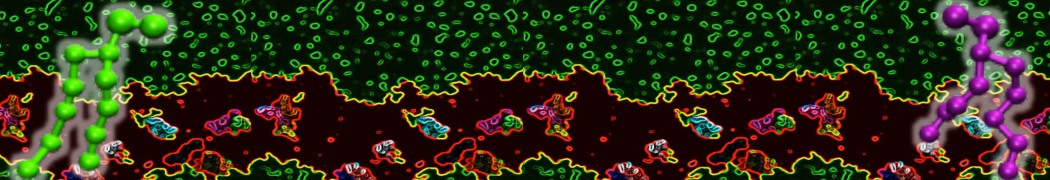
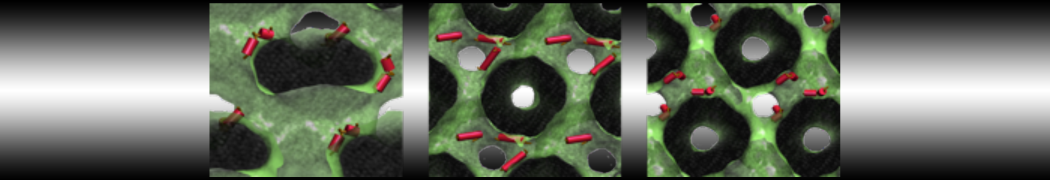
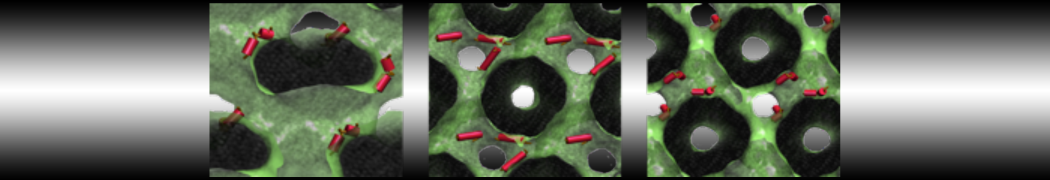
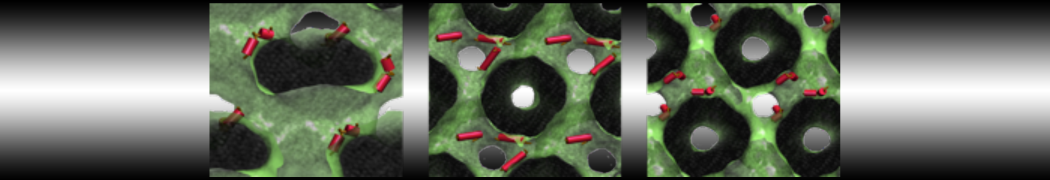
My water is freezing, help!
The unwanted freezing of water is a known problem in the current parameterization (version 2). It has already been observed and discussed in our previous work [1,2,3]. Please note the following points:
i) although the LJ parameters for water (ε=5.0 kJ mol-1, σ=0.47 nm) bring it into the solid state region of the LJ phase diagram, the use of a shift potential reduces the long-range attractive part. Consequently, the CG water is more fluid compared to the standard LJ particle.
ii) We have previously [2] determined the freezing temperature of the CG water as 290 +/- 5K. While this is admittedly higher than it should be, in most applications freezing is not observed as long as no nucleation site is formed. Apart from simulations performed at lower temperatures, rapid freezing is therefore a potential problem in systems where a nucleation site is already present (a solid surface, but also an ordered bilayer surface may act as one) or when periodicity enhances the long range ordering (e.g., for small volumes of water).
iii) In those cases in which the freezing poses a problem, a simple pragmatic solution has been presented in the form of antifreeze particles. This works in some cases, but has apparently given problems in combination with solid supports. Therefore, be careful to check that your antifreeze particles do not cluster. You may also switch to the polarizable water model which has a lower melting temperature [4].
Anyhow, the freezing of Martini water is clearly not ideal, and there is room for improvement. In future versions of the MARTINI force field we intend to use a softer potential with a tuneable width to extend the fluid range of the water model.
[1] S.J. Marrink, H.J. Risselada, S. Yefimov, D.P. Tieleman and A.H. de Vries. The MARTINI force field: Coarse grained model for biomolecular simulations. J. Phys. Chem. B, 2007, 111, 7812-7824.
[2] S.J. Marrink, A.H. de Vries and A.E. Mark, Coarse grained model for semiquantitative lipid simulations, J. Phys. Chem. B, 2004, 108, 750–760.
[3] S.J. Marrink, X. Periole, D.P. Tieleman, A.H. de Vries. Comment on using a too large integration time step in molecular dynamics simulations of coarse-grained molecular models. Phys. Chem. Chem. Phys., 12:2254-2256, 2010. abstract
[4] S.O. Yesylevskyy, L.V. Schäfer, D. Sengupta, S.J. Marrink. Polarizable water model for the coarse-grained Martini force field. PLoS Comp. Biol, 6:e1000810, 2010. open access
My protein starts deforming, help!
Time to use an elastic network in combination with your Martini protein. See the ELNEDYN tutorial page.
My vesicles do not want to fuse, help!
Well, some vesicles simple do not like to fuse so maybe your expectations should be lowered ..... however, there could be a potential artificial barrier toward fusion if you use Martini 2.0 or higher. In order to make the solvation free energy of ions in apolar media more realistic, in version 2.0 the interaction between Q type particles and C1/C2 particles was changed to the 'super-repulsive' category. Here the effective size of the Q particles (or C particles, depending on your perspective) was increased to 0.6 nm, much larger than the standard interaction size of 0.47 nm. This effectively blocked ions from dissolving into apolar solvents such as the bilayer interior. However, it also has the unwanted effect that it hinders lipid tails from protruding toward and through the lipid/water interface. The first barrier to vesicle fusion is the formation of a stalk, which is triggered by the protrusion of lipid tails ...
In order to remove this artefact, simply change the Q-C1/C2 interactions back to their normal 'repulsive' interaction style, but do realize that this will also affect the ability of ions to pass through membranes. If you want to look at fusion in the presence of ions, you might want to add a different particle type to distinguish the Q site of the lipid headgroup versus the Q sites of the ions ..
See also:
[1] D. Mirjanian, A.N. Dickey, J.H. Hoh, T.B. Woolf, M.J. Stevens. Splaying of Aliphatic Tails Plays a Central Role in Barrier Crossing During Liposome Fusion. J. Phys. Chem. B 2010, 114, 11061–11068, in which this trick was applied.
I cannot log-in or post on the forum!
To be able to post on the Martini forum, you have to log-in: From the "User Platform" menu, choose the "Login to Forum". To be able to log-in here, you have to be a registered user. Click the "Register" link at the bottom of the page and provide the required personal data. An e-mail containing an activation link will be sent to the address provided. Check your spam folder in case you did not receive an email!
Who can I contact with further questions?
In case you do not find your answer anywhere (FAQ, user platform, tutorial session ...), or if you experience unreported problems, please send an email to:
This email address is being protected from spambots. You need JavaScript enabled to view it.