- Posts: 2
 unstable protein-peptide binding with Martini 3
unstable protein-peptide binding with Martini 3
- qiaobf
-
 Topic Author
Topic Author
- Offline
- Fresh Boarder
Less
More
2 years 9 months ago #9165
by qiaobf
unstable protein-peptide binding with Martini 3 was created by qiaobf
Hi all,
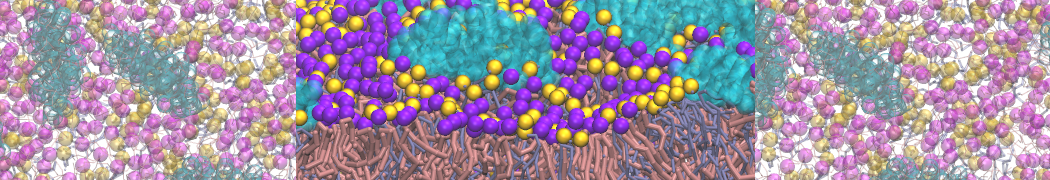
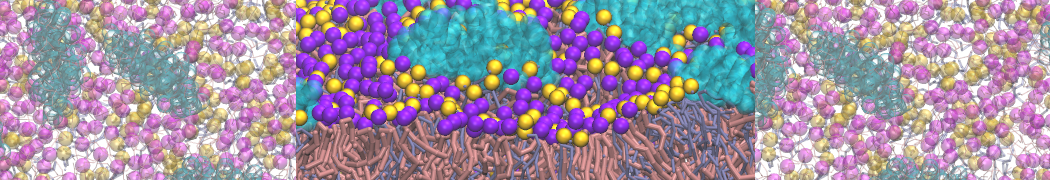
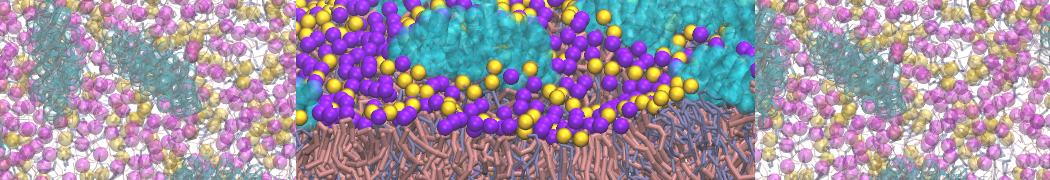
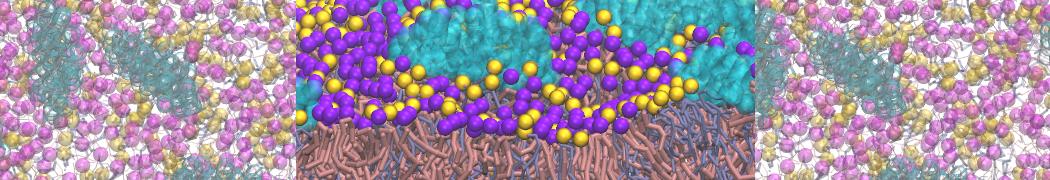
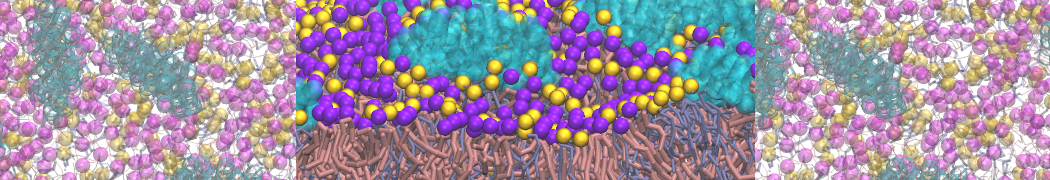
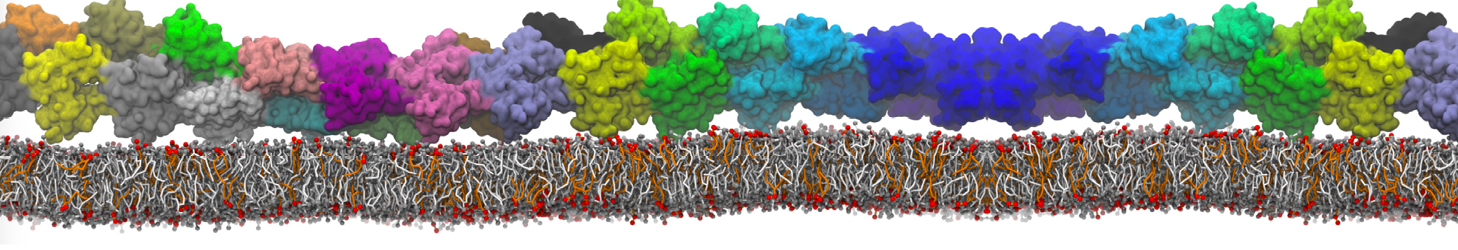
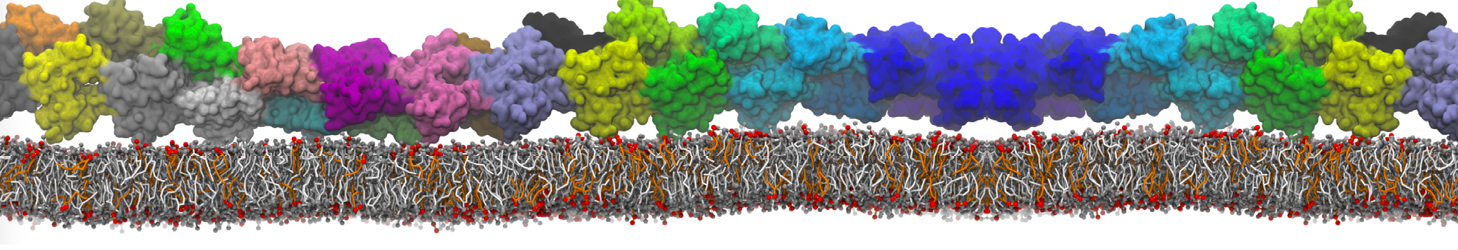
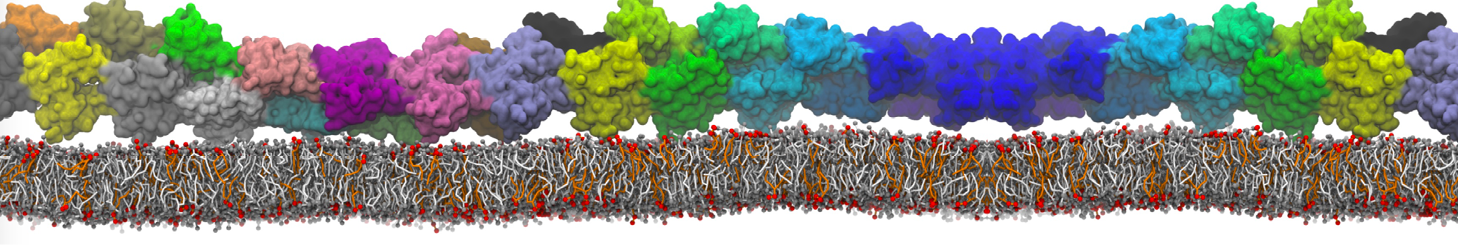
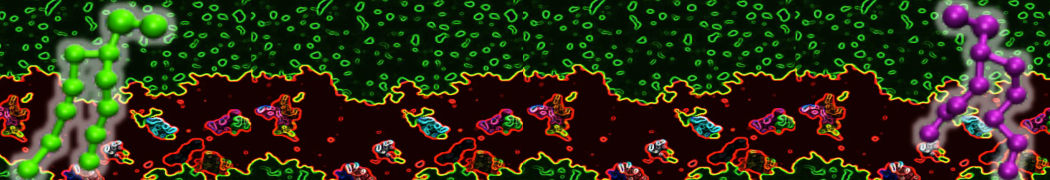
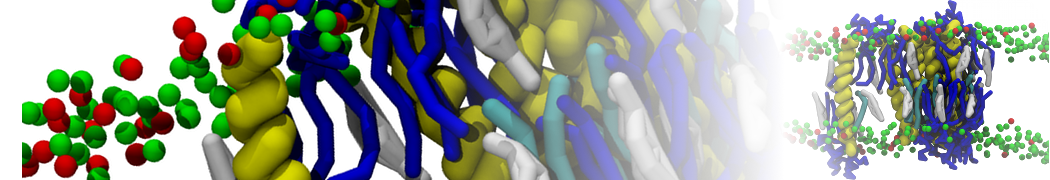
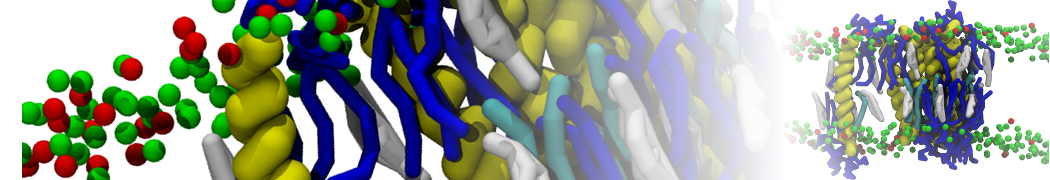
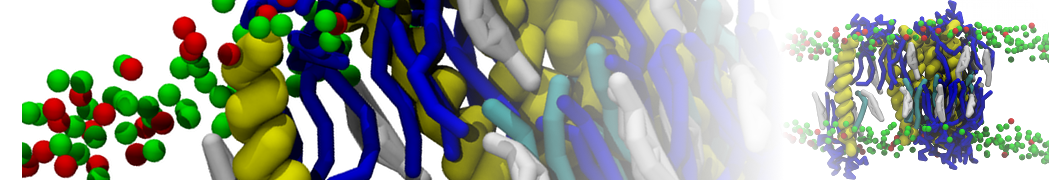
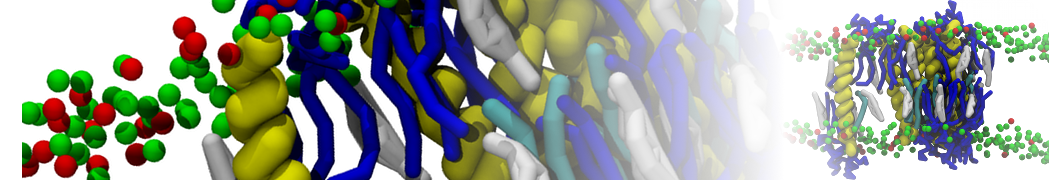
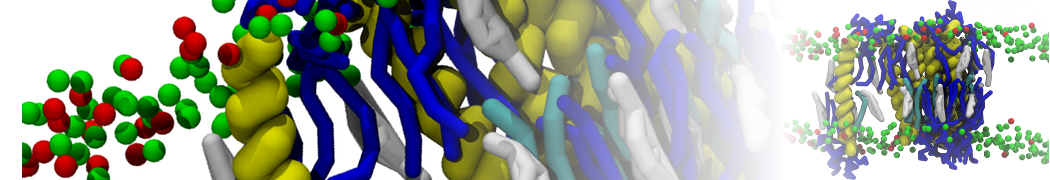
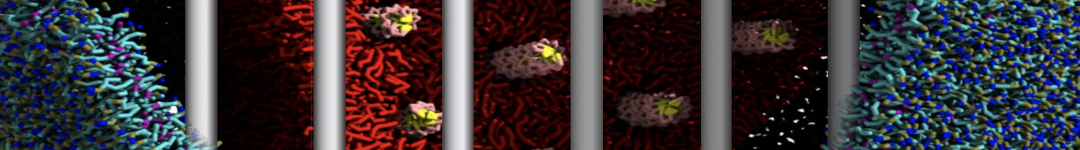
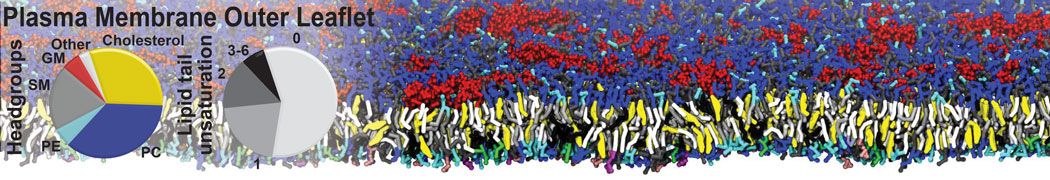
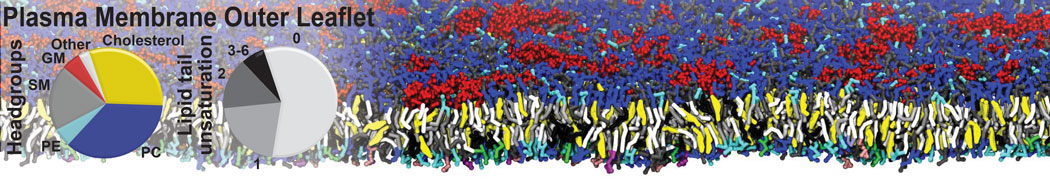
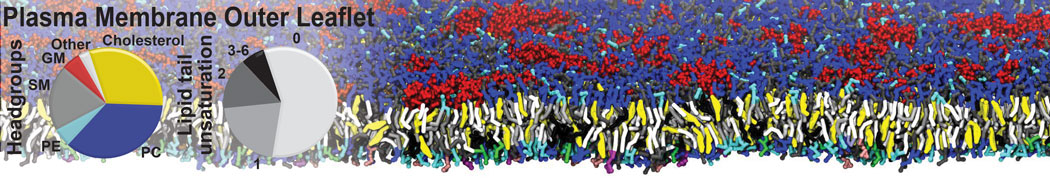
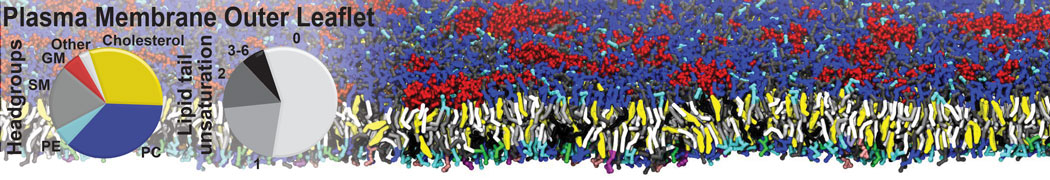
It is great to see the release of the MARTINI3. I am planning to apply it on one protein-peptide binding study, but found that the protein-peptide binding structure is unstable with MARTiNI3 (which is stable for Martini 2.2 and CHARMM). Does anyone have any suggestions or comments? Sincerely appreciate your kind help.
What I did is following.
1) Download the crystal structure from rcsb.org
2) generate the all-atom structure (by adding hydrogen using gmx pdb2gmx)
3) Map all-atom structure to Martini CG structure
(3.a) Protein:
martinize2 -f protein_AA.pdb -ff martini3001 -x protein_CG.pdb -o system.top -dssp -cys auto -elastic -scfix -p backbone -maxwarn 1
(3.b) Peptide:
martinize2 -f peptide_AA.pdb -ff martini3001 -x peptide_CG.pdb -o system.top -dssp -cys auto -elastic -scfix -p backbone -nt
(4) merge the CG structures, dissolve the protein-peptide complex in water, add 0.15 M NaCl.
(5) equilibrate the simulation box:
(5.a) energy minimization;
(5.b) NVT with position restraints on Protein+peptide (POSRES_FC=1000), 10ns;
(5.c) NPT with position restraints on Protein+peptide (POSRES_FC=1000), 100ns;
(5.d) NPT with position restraints on Protein+peptide (POSRES_FC=100), 100ns;
(5.e) NPT with position restraints on Protein+peptide (POSRES_FC=10), 100ns;
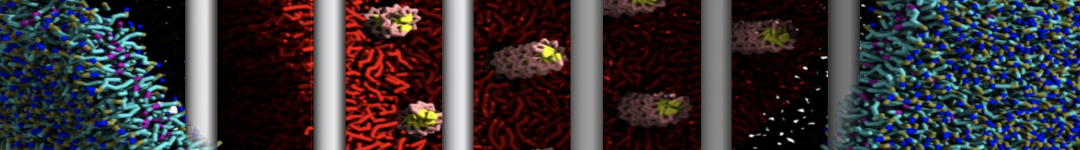
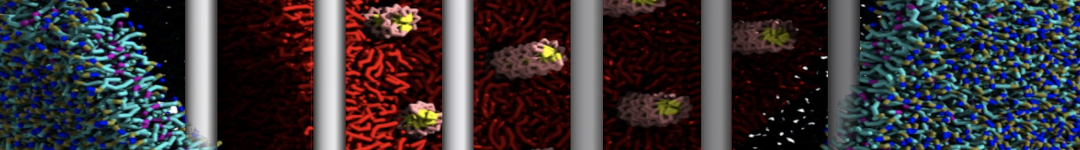
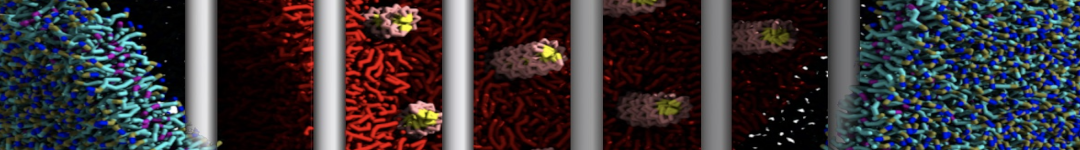
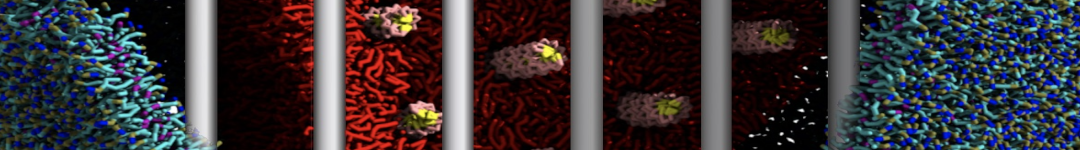
(6) run the production simulation (no position restraints). But the protein-peptide complex became disassociated quickly. More surprisingly, the protein-peptide binding is not stable at all, which is disassociated for most of the simulation time, and occasionally forming binding structure for a short period. (In contrast, the protein-peptide complex is very stable using the MARTINI 2.2 potential and the CHARMM all-atom potential) The peptide is seemingly much more polar in the MARTINI 3 potential.
Additionally I did two tests. Unfortunately, I got similar results that the protein-peptide complex is not stable.
(a) peptide with the backbone type of P5 (instead of the default P6) for the charged N- and C-terminal groups;
(b) peptide with charged N- and C-terminal groups (martinize2 -f peptide_AA.pdb -ff martini3001 -x peptide_CG.pdb -o system.top -dssp -cys auto -elastic -scfix -p backbone).
Sincerely appreciate your time reading this long email, and kind help.
It is great to see the release of the MARTINI3. I am planning to apply it on one protein-peptide binding study, but found that the protein-peptide binding structure is unstable with MARTiNI3 (which is stable for Martini 2.2 and CHARMM). Does anyone have any suggestions or comments? Sincerely appreciate your kind help.
What I did is following.
1) Download the crystal structure from rcsb.org
2) generate the all-atom structure (by adding hydrogen using gmx pdb2gmx)
3) Map all-atom structure to Martini CG structure
(3.a) Protein:
martinize2 -f protein_AA.pdb -ff martini3001 -x protein_CG.pdb -o system.top -dssp -cys auto -elastic -scfix -p backbone -maxwarn 1
(3.b) Peptide:
martinize2 -f peptide_AA.pdb -ff martini3001 -x peptide_CG.pdb -o system.top -dssp -cys auto -elastic -scfix -p backbone -nt
(4) merge the CG structures, dissolve the protein-peptide complex in water, add 0.15 M NaCl.
(5) equilibrate the simulation box:
(5.a) energy minimization;
(5.b) NVT with position restraints on Protein+peptide (POSRES_FC=1000), 10ns;
(5.c) NPT with position restraints on Protein+peptide (POSRES_FC=1000), 100ns;
(5.d) NPT with position restraints on Protein+peptide (POSRES_FC=100), 100ns;
(5.e) NPT with position restraints on Protein+peptide (POSRES_FC=10), 100ns;
(6) run the production simulation (no position restraints). But the protein-peptide complex became disassociated quickly. More surprisingly, the protein-peptide binding is not stable at all, which is disassociated for most of the simulation time, and occasionally forming binding structure for a short period. (In contrast, the protein-peptide complex is very stable using the MARTINI 2.2 potential and the CHARMM all-atom potential) The peptide is seemingly much more polar in the MARTINI 3 potential.
Additionally I did two tests. Unfortunately, I got similar results that the protein-peptide complex is not stable.
(a) peptide with the backbone type of P5 (instead of the default P6) for the charged N- and C-terminal groups;
(b) peptide with charged N- and C-terminal groups (martinize2 -f peptide_AA.pdb -ff martini3001 -x peptide_CG.pdb -o system.top -dssp -cys auto -elastic -scfix -p backbone).
Sincerely appreciate your time reading this long email, and kind help.
Please Log in or Create an account to join the conversation.
Time to create page: 0.092 seconds